Orion Cortical Visual Prosthesis System gets FDA Expedited Access Pathway designation
What is Orion Cortical Visual Prosthesis System?
Orion Cortical Visual Prosthesis System was developed by Second Sight Medical Products, Inc. , the same company that manufactured Argus II Retinal Prosthesis System. The Argus II was designed to help patients with retinitis pigmentosa—a genetic disorder characterized by loss of cells in the retina—to regain some of the lost vision.
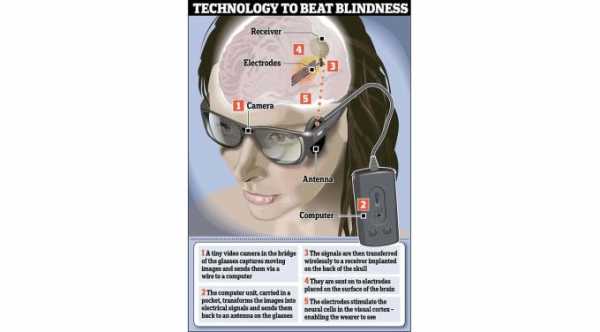
The Argus II captures images with the use of a very small video camera attached to a patient’s Eyeglasses. The images are converted into electrical pulses which are wirelessly transmitted to special electrodes that are implanted on the surface of the retina.
These electrical pulses stimulate the remaining cells in the retina to create visual patterns for the patient. The Argus II was the first artificial retina that received commercial approval.
The concept behind Orion is similar to Argus II, wherein a small video camera is attached to the patient’s eyeglasses to capture the images. The images are then converted into electrical pulses that are sent wirelessly to special electrodes attached on the surface of the visual cortex—the portion of the brain that receives and processes impulses from the optic nerve.
Orion bypasses the retina and the optic nerve and has the potential to restore vision to blind patients who lost their vision due to a variety of different conditions.
Orion Cortical Visual Prosthesis System has been awarded Expedited Access Pathway (EAP) designation by the U.S. Food and Drug Administration (FDA). The said designation which is awarded to certain medical devices allows reduced time and cost for development, priority review, and potential approval from the FDA to expedite its availability in the market. This means that patients will have a faster access on Orion.
An EAP designation is given to selected medical devices that have the potential to treat life threatening or irreversibly debilitating conditions which are subject to pre-market approval applications, premarket notification or requests for De Novo designation.
The EAP may allow the FDA to approve Orion even with fewer patients and lesser follow-up time frame. According to the FDA, all medical devices with an EAP designation will achieve a Breakthrough Device designation when the guidance document is finalized.
As for Orion, a draft guidance document for the Breakthrough Device Program has also been released by the FDA. Once finalized, Orion’s EAP designation will be replaced with a Breakthrough Device designation.
Advantages over the review process of FDA:
1.Better interactive review for Investigational Device Exemption and Premarket Approval application.
2.Greater reliance on post-market vs. pre-market data collection.
3.Greater acceptance of uncertainty in the benefit-risk profile during approval.
4.Review of the submission is prioritized on the review queue with additional review resources provided.
5.Involvement of the senior FDA management.
Second Sight Medical announced the first human trial implantation of Orion Cortical Visual Prosthesis System in late January. It was performed by Dr. Nader Pouratian, Vice Chairman of the Department of Neurosurgery at the Ronald Reagan UCLA Medical Center (UCLA). The company is expecting additional four patients to be enrolled in the study.




