Macular Dystrophy Treatment in Stargardt Disease
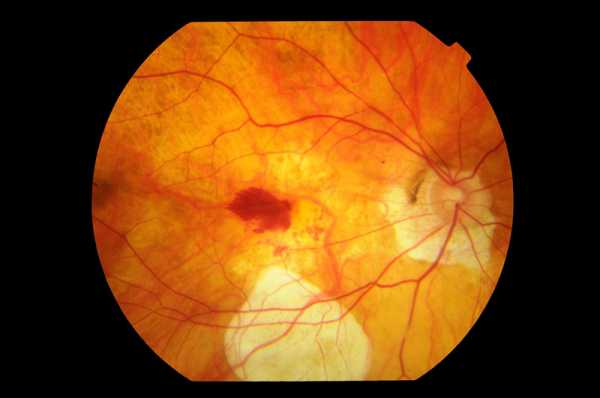
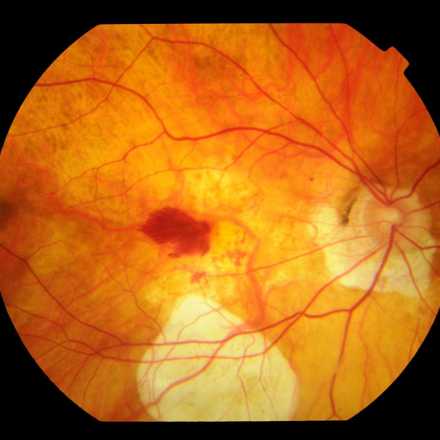
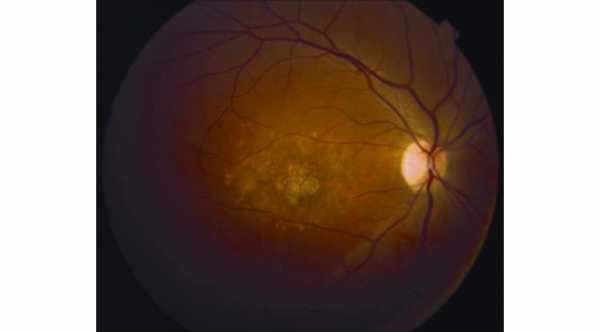
Stargardt disease is one of the most common macular dystrophy or degeneration in the world that affects children and young adults between the ages of 10 to 20 years.
This Disease affects the photoreceptors and retinal pigment epithelium layers which will lead to progressive vision loss. So far, there is no treatment or cure for Stargardt's disease.
A new treatment has emerged out but it still under clinical trials. This new treatment involves embryonic stem cells.
These stems cells will be transformed into retinal pigment epithelial cells. These new healthy retinal pigment epithelial cells will be transplanted into the subretinal area in patients with advanced Stargardt macular dystrophy.
Retinal pigment epithelial cells have many essential functions and one of them is to support photoreceptor cells which are responsible for vision.
Advanced Cell Technology (ACT) is a Biotechnology company and this company is involved in these clinical trials. It received authorization from the FDA to begin human clinical trial of Stargardt macular dystrophy treatment derived from human embryonic stem cells.
ACT also is planning to extend this clinical trial on patients with other types of macular dystrophy such as Age Related Macular Degeneration.